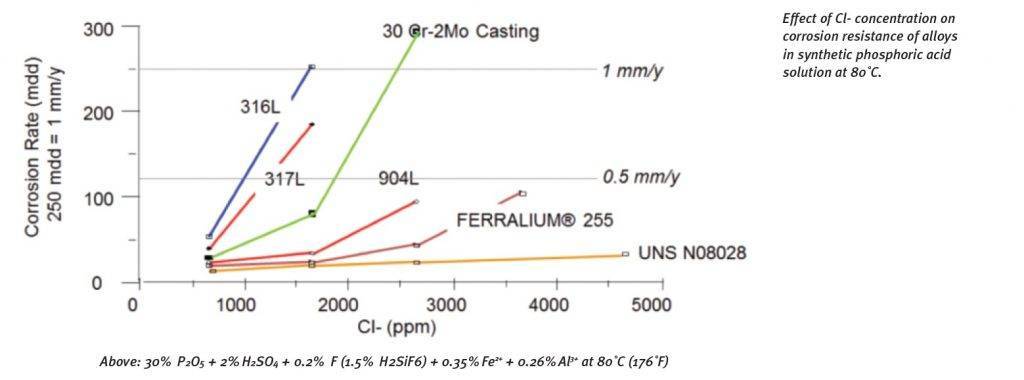
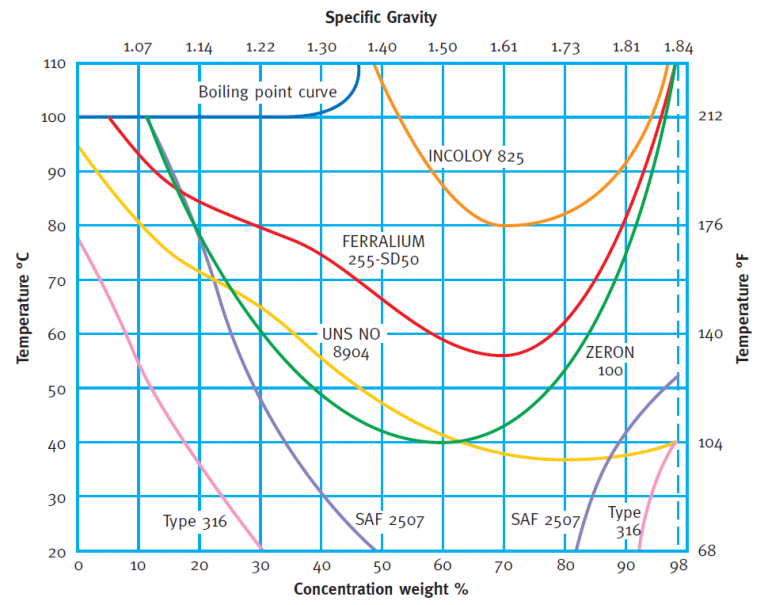
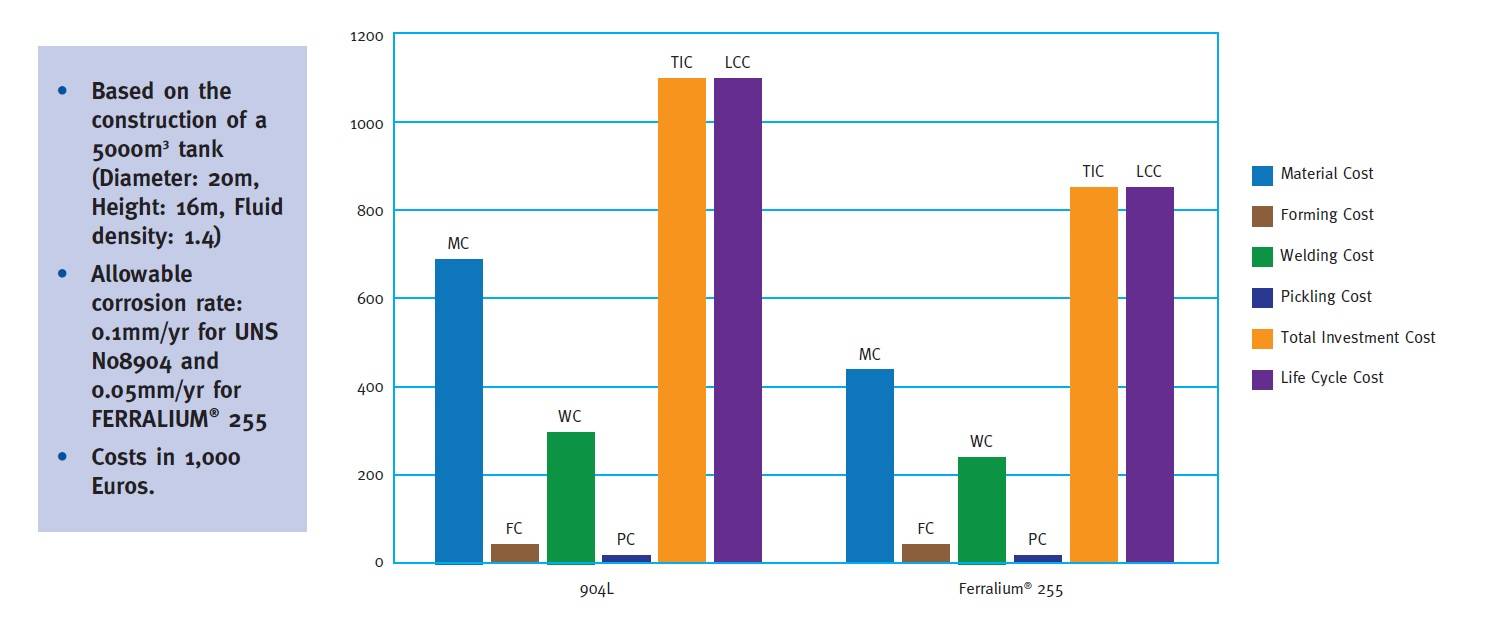
Phosphoric Acid based Fertiliser Production
The Chemical Processing Industry (CPI) encompasses a broad range of products, including petrochemical and inorganic chemicals, plastics, detergents, paints, pigments and more. Given the specific nature of individual processes, we focus here on the production of phosphoric acid – a key precursor in the large-scale production of fertilisers and agri-chemicals – but there is some commonality between the process conditions experienced in other acid-based processes.